Energy of AtomĪbsorption Spectra:An atom obtains a higher energy level when an absorption spectrum is given by that atom.Įmission Spectra: An emission spectrum is given when an excited atom obtains a lower energy level. AppearanceĪbsorption Spectra: Absorption spectra show dark lines or gaps.Įmission Spectra:Emission spectra show colored lines. Energy ConsumptionĪbsorption Spectra:An absorption spectrum is produced when atoms absorb energy.Įmission Spectra:An emission spectrum is produced when atoms release energy. 
This indicates that an absorption spectrum is specific to a particular atom.įigure 2: Emission Spectrum of Helium Difference Between Absorption and Emission Spectra DefinitionĪbsorption Spectra:An absorption spectrum can be defined as a spectrum obtained by transmitting electromagnetic radiation through a substance.Įmission Spectra:Emission spectrum can be defined as a spectrum of the electromagnetic radiation emitted by a substance. 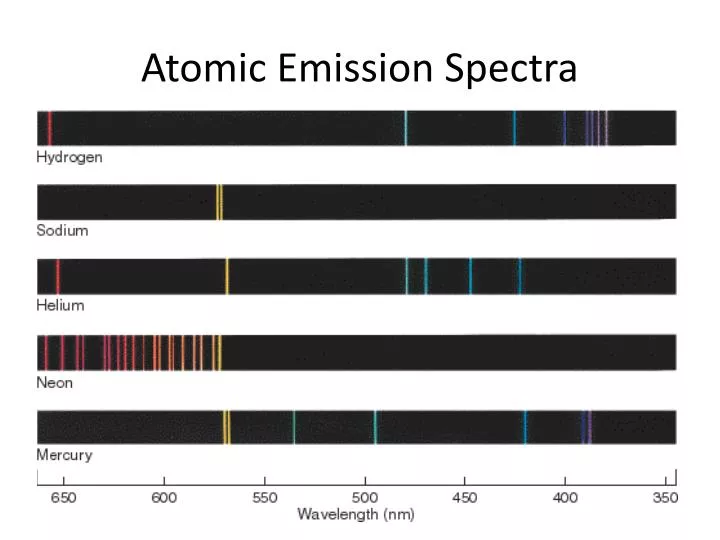
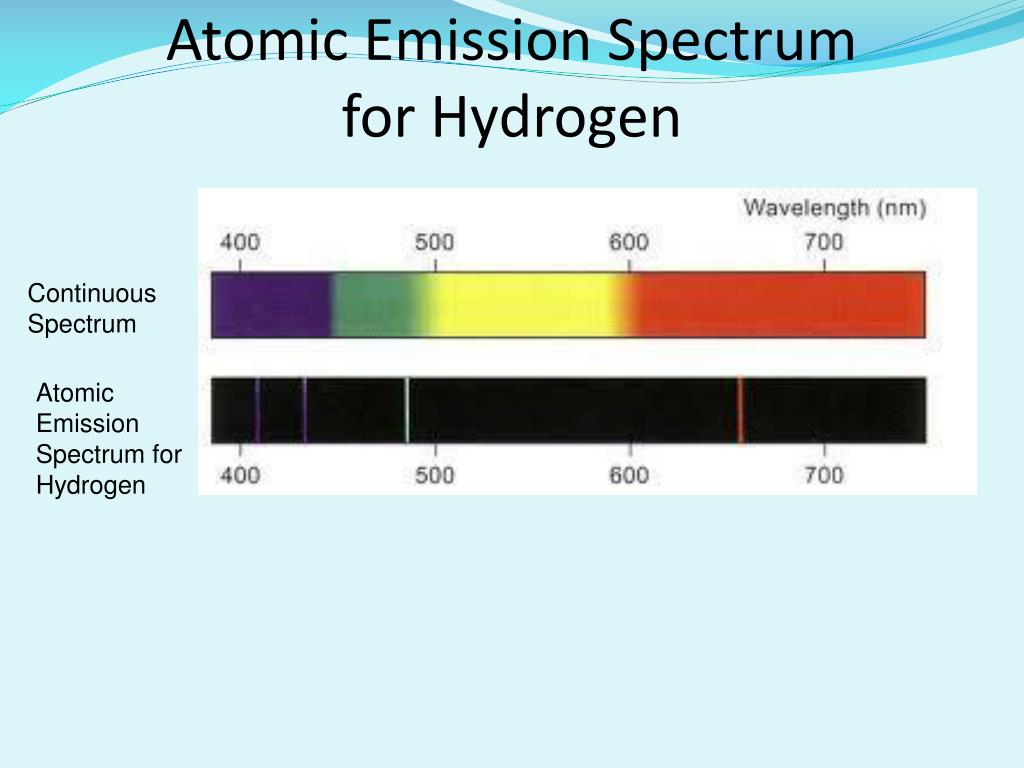
The energy levels and their locations in an atom can also be determined from this. Since the continuous spectrum of the light source is given as the wavelength range of electromagnetic radiation, the missing wavelengths can be found. Therefore, the energy is inversely proportional to the wavelength of the electromagnetic radiation. H – Plank’s constant (Js) λ – Wavelength (m) Where, E – energy of the photon (Jmol -1) c – Speed of radiation (ms -1) Then the transmission of the radiation through the substance gives colored bands that correspond to the photons that were not absorbed dark lines indicate the photons which were absorbed. But if the energy of the photon is not equal to the energy difference between two energy levels, the photon is not going to be absorbed. Thus, it jumps to the higher energy level. Then the energy of that electron is high. This absorption causes the energy of that particular electron to be increased. If the energy of the photon is the same as the energy between two energy levels, then the photon’s energy is absorbed by the electron in the lower energy level. When a substance is exposed to an electromagnetic radiation source such as white light, it can obtain the absorption spectra. The characteristic feature of an absorption spectra is that it shows dark lines on the spectrum.Ībsorption spectrum is a result of absorbing photons by the atoms present in the substance. Key Terms: Atom, Absorption Spectra, Emission Spectra, Orbital, Photon, Shell What are Absorption SpectraĪn absorption spectrum can be defined as a spectrum obtained by transmitting electromagnetic radiation through a substance. What is the difference between Absorption and Emission Spectra The main difference between absorption and emission spectra is that absorption spectra show black colored gaps/lines whereas emission spectra show different colored lines in the spectra. Both absorption and emission spectra are line spectra. These movements can be used to obtain absorption and emission spectra. When energy is given to an atom externally, it causes the electrons to jump from one shell to another. The shell which is the nearest to the nucleus is known to have the lowest energy. According to the modern atomic theory, these electrons are positioned in specific energy levels called shells or orbitals where their energies are quantized. The structure of an atom includes a central core called a nucleus and a cloud of electrons around the nucleus. Main Difference – Absorption vs Emission Spectra
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
